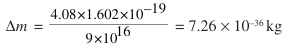chemical reactions also conform to E = mc²
April 16, 2026
This was a 12th grade Gestalt moment for me, of course ages ago: Even in ‘Chemical’ reactions, the mass-energy equivalence applies, like mass-defect in the Nuclear ones.
Chemical reaction energies (endothermic/exothermic) arise from electronic interactions++; the associated mass defect is a secondary consequence.
E=mc² For The Win. YES!
1
Many of us erroneously assume that Chemical energy creation (or absorption) does not involve anything like mass-energy equivalence; and that, ‘masses are like totally conserved in chemical reactions.’ That, the ‘sum of the masses of reactants on the left hand side of an equation, are equal to the products/results on the right hand side.’
Our school textbooks (hic) feed the ignorance by creating random differences between nuclear and chemical processes/reactions – which our otherwise mediocre ‘teachers’ follow like automatons; of course, there are obvious differences though.
(having been a school-teacher before – and sorta continuing as a low intensity one-on-one teacher/cheater for a while, and stressing on the idea ad infinitum – I can say with a huge satisfaction that many of my young learners have had such similar gestalt moments of wonder that is Science!)
2
FACT: Even the burning of firewood has to adhere to the stellar idea of Mass-Energy equivalence as fantastically propounded by Albert Einstein.
The basic interaction/reaction in ‘burning firewood resulting in smoke & heat/light’ is of carbon atoms combining with oxygen molecules to produce carbon dioxide:
C + O2 → CO2 + 4.08 eV.
Remember: 1 electron volt (eV) = ~1.6 × 10⁻¹⁹ joule (J) = ~3.83 x 10-20 calorie. A calorie is nothing but the heat energy required to increase the temperature of 1 gram of water by 1 degree centigrade; of course 1 eV has a very, very small or insignificant heat equivalent – but, the fact is that it exists.
Anyway. In this exothermic (heat releasing) chemical reaction, 4.08 eV of energy is released for each reacting carbon atom; it is referred to as the binding energy (BE) of CO2 molecule.
Again, as a reverse reaction – if 4.08 eV of energy is given to a CO2 molecule under suitable conditions, it will break up into its constituents as in:
CO2 + 4.08 eV → C + O2
… what this means is 4.08 eV energy leaves the system to form CO2 gas. Therefore, the mass of CO2 molecule will be less than the total mass of C and O2 by a mass equivalent of 4.08 eV.
This loss of mass Δm can be calculated using the brilliant equivalence relation E = mc2 …
Of course, such a small change in mass cannot be detected and we say without batting an eyelid that the mass is conserved in chemical reactions, though slight change of mass does occur.
(In one gram of Carbon, there are ~5.02 × 1022 atoms; when one gram of Carbon is burnt thus, the heat released is 7800 calories)
3
Summarizing various aspects of chemical reactions…
- Energies of the order of 10 eV are involved. (in Nuclear ones, the order is in MeV – million/mega eVs = 106 eVs)
- Change of mass is of the order of 10–35 kg, which is extremely small and so we claim that the mass is conserved.
- The total number of atoms of each type on the right hand side of a given chemical equation is always equal to the total number of atoms of each type on the left hand side.
- Chemical energy comes from electronic structure changes (rearrangement of valence electrons, bond formation/breaking) – & is governed by electromagnetic interactions
- The mass change is a derived consequence, not the driver
- In a chemical bond – the electrons rearrange into a more stable (lower-energy) configuration
E=mc² For The Win. Absolutely YES!
END